One year when I had passed kidney stones the doc gave me sulfer pills to ease the pain of post-tract shredage where one was extra misbehaved on the way out. The bright orange dye definitely marks your territory.Asparagus is my fav food for aromatic urine. I will go around and mark my property corners after feasting
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Dehydration thread
- Thread starter bykfixer
- Start date
 Help Support Candle Power Flashlight Forum
Help Support Candle Power Flashlight Forum
This is what is left of the pea soup I made on Monday.
The picture is under fluorescent lighting.

The picture is under fluorescent lighting.
electrolyte
Newly Enlightened
- Joined
- Jan 4, 2019
- Messages
- 61
Forgive me for not reading the whole thread. I never thought much of drinking sodium chloride solutions. It seems like a bad idea. I used to ride in the mountains outside of Tucson twice a week, 50 miles a pop in ~2.5h. I'd put away over 7 l of water on some days. I always, however, had a pocket full of grapes, bananas or some combination of the two. There is lots of potassium there and the calories were going out as fast as the water.
- Joined
- Mar 4, 2012
- Messages
- 643
Forgive me for not reading the whole thread. I never thought much of drinking sodium chloride solutions. It seems like a bad idea. I used to ride in the mountains outside of Tucson twice a week, 50 miles a pop in ~2.5h. I'd put away over 7 l of water on some days. I always, however, had a pocket full of grapes, bananas or some combination of the two. There is lots of potassium there and the calories were going out as fast as the water.
Things are not always as they "seem".
Salt is by far the highest component of minerals lost due to sweat. Potassium takes many hours for uptake in the body, so eating anything for potassium does not work in real time to replace electrolytes. It literally takes hours to days to raise potassium levels. Salt, however, is very, very quickly absorbed by the body. So your technique is opposite of what actually works for quickly replacing electrolytes lost due to sweat.
There is some merit to eating grapes during exercise, as they are mostly water and distribute fluid over time for rehydration. Bananas aren't quite as good, but they are also about 75% water. If you ate the fruit for that purpose, it probably helped. Plus over time your potassium levels stayed high, but you could have eaten them hours before, during, or hours after exercise for the same effects, unlike a salt solution, which helps very quickly (literally in minutes).
REFERENCE (one of many you can find with a search on NIH.gov)
"Key Points. Eating up to 2 servings of bananas caused marginal increases in plasma potassium concentration. The small increases in plasma potassium concentration occurred 30 to 60 minutes postingestion of bananas. Eating bananas is unlikely to be an effective treatment for exercise-associated muscle cramping."
NIH Study of Potassium Replacement
Last edited:
After a regiment of 2 litres of water a day I was still getting side stitch muscle cramps and shriveled skin. Sports drinks just passed through me. So each day I eat a fruit or vegetable of some sort that is mostly water like cantelope or grapes, and nuts that are high in magnesium like cashews or almonds. A cup of yogurt, some carrots, brocoli, peanuts etc along with my 2 liters of water and so far so good.
It seems by getting electrolites through food and hydration from water it's a pretty good balance. When I sweat a lot I add more water, eat bananas and grapes etc too. Trouble is it's too easy to not consume the correct foods when meals are rushed due to fast past, hectic lifestyles.
It seems by getting electrolites through food and hydration from water it's a pretty good balance. When I sweat a lot I add more water, eat bananas and grapes etc too. Trouble is it's too easy to not consume the correct foods when meals are rushed due to fast past, hectic lifestyles.
+
Good overall view & approach. So much in life is 'balance'
I keep a gallon of water next to my bed and drink a fair amount every night.
If nature calls during the night, can think of worse things.
+After a regiment of 2 litres of water a day I was still getting side stitch muscle cramps and shriveled skin. Sports drinks just passed through me. So each day I eat a fruit or vegetable of some sort that is mostly water like cantelope or grapes, and nuts that are high in magnesium like cashews or almonds. A cup of yogurt, some carrots, brocoli, peanuts etc along with my 2 liters of water and so far so good.
It seems by getting electrolites through food and hydration from water it's a pretty good balance. When I sweat a lot I add more water, eat bananas and grapes etc too. Trouble is it's too easy to not consume the correct foods when meals are rushed due to fast past, hectic lifestyles.
Good overall view & approach. So much in life is 'balance'
I keep a gallon of water next to my bed and drink a fair amount every night.
If nature calls during the night, can think of worse things.
electrolyte
Newly Enlightened
- Joined
- Jan 4, 2019
- Messages
- 61
The amounts of sweat and the amount of salt in sweat vary a great deal between individuals. The question of how much sodium you are losing in heavy exercise is beyond what just about everyone can determine because we don't have access to the analytical tools to find out.
Most people in our country consume as much or way more sodium than is needed and recommended. It contributes to health problems. If you have high blood pressure, you should be very careful about intentionally adding salt to your diet. Get some professional advice.
If you are exercising for less than 45 minutes (maybe less than 30 in very hot and humid weather, you are unlikely to need sodium supplementation.
Low blood sodium levels can be uncomfortable and dangerous, but people that experience this as a result of vigorous exercise are as likely to have precipitated their problem by drinking too much water as by avoiding sodium chloride. This is easy to get at, weigh yourself before and after exercise and if you weigh more after, you likely drank more than you need.
One thing for sure is that drinking cold water is not a good way to absorb it fast. If you are sipping cold water, that is fine since it won't cool you innards that way, just don't gulp cold water.
Water amounts, sodium amounts and sodium-potassium ratio are all important, but a lot more needs to be done. Even after that, individual variation means that everyone will still likely be a black box.
Most people in our country consume as much or way more sodium than is needed and recommended. It contributes to health problems. If you have high blood pressure, you should be very careful about intentionally adding salt to your diet. Get some professional advice.
If you are exercising for less than 45 minutes (maybe less than 30 in very hot and humid weather, you are unlikely to need sodium supplementation.
Low blood sodium levels can be uncomfortable and dangerous, but people that experience this as a result of vigorous exercise are as likely to have precipitated their problem by drinking too much water as by avoiding sodium chloride. This is easy to get at, weigh yourself before and after exercise and if you weigh more after, you likely drank more than you need.
One thing for sure is that drinking cold water is not a good way to absorb it fast. If you are sipping cold water, that is fine since it won't cool you innards that way, just don't gulp cold water.
Water amounts, sodium amounts and sodium-potassium ratio are all important, but a lot more needs to be done. Even after that, individual variation means that everyone will still likely be a black box.
^^ Well put
Years ago I worked with a really talented young engineer who always drank his water warm. I asked why. He said when it enters your body at the same temperature as your body no energy is required to cool it or warm it. True or not? I dunno, it made sense to me so I usually drink room temperature water to rehydrate.
Years ago I worked with a really talented young engineer who always drank his water warm. I asked why. He said when it enters your body at the same temperature as your body no energy is required to cool it or warm it. True or not? I dunno, it made sense to me so I usually drink room temperature water to rehydrate.
Last edited:
PhotonWrangler
Flashaholic
Interesting. I usually drink my electrolyte water at room temperature, unless it's one of those flavors that has to be chilled or it tastes hideous.Years ago I worked with a really talented young engineer who always drank his water warm. I asked why. He said when it enters your body at the same temperature as your body no energy is required to cool it or warm it. True or not? I dunno, it made sense to me so I usually drink room temperature water to rehydrate.
- Joined
- Mar 4, 2012
- Messages
- 643
^^ Well put
Years ago I worked with a really talented young engineer who always drank his water warm. I asked why. He said when it enters your body at the same temperature as your body no energy is required to cool it or warm it. True or not? I dunno, it made sense to me so I usually drink room temperature water to rehydrate.
That is true, but has nothing to do with why warm water might be better than very cold (or hot). Yes, I am an engineer (MSME Georgia Tech).
Folks just tend to drink more room temperature or slightly chilled water than very cold or very warm when they are dehydrated. So you will likely rehydrate faster from room temp or slightly cooled water (because you drink more).
How much energy does it take to raise an 8oz glass of water from 39 degrees (fridge temperature) to 98.6 degrees? Only about 8 calories!
Jean-Luc Descarte
Enlightened
In the tropics, "room temperature" water means lukewarm water. I only drink mine chilled or icy.
At work there is a huge industrial-grade drinking fountain that dispenses water cold enough to fog up the tap and cause brain freeze if you drink too fast. It was inoperable for a few weeks last year, and in retrospect that coincided with me urinating orange juice day in and day out. Most of my hydration comes from that thing.
At work there is a huge industrial-grade drinking fountain that dispenses water cold enough to fog up the tap and cause brain freeze if you drink too fast. It was inoperable for a few weeks last year, and in retrospect that coincided with me urinating orange juice day in and day out. Most of my hydration comes from that thing.
electrolyte
Newly Enlightened
- Joined
- Jan 4, 2019
- Messages
- 61
The engineer's reasoning has little to do with heat-stressed athletes. That's the quoted engineer, not Jim. People do tend to be able to drink more cool water than cold water when under heat stress. Think about it, can any of you chug lots of truly cold fluids if they don't contain anesthetic alcohol?
Let's round things a little and say that body temp is 100F and you are considering drinking water at 40F or 70F. Water has much of the heat capacity in a body. The other stuff has some, but not nearly as much as water which is one of the unusual qualities of that substance. A person of 167# has 100# of water. If your 167# drinks a pint of 40F or 70F of water, you will lower your body temp by roughly 0.6 or 0.3 degrees respectively assuming total and immediate "mixing" That is much more significant that I might have assumed before looking at it quantitatively.
I am however, disturbed by the lack of detail that I find in discussions by people who project as experts in this respect. We are not just a bag of water and some other stuff being diluted with a little bag of water.
I have a vague recollection of some expert stating that drinking cold water slows absorption. It makes sense that cold water makes your gut cold and slows everything down at best and shuts it down at worst. I just went with that in my previous post, shame on lazy me. Local drastic cooling, however, just slows or stops everything that an organ is doing and that includes absorbing water. In a biology laboratory, we make processes of all kinds stop, or nearly stop by putting them in and ice-water bath. (That said, it is not as effective with poiklothermic cells/tissues/animals.)
I am not trying to refute that "cold stunned gut" reasoning, but I find little data to support it. There is data that supports drinking cold or cool water on the one hand, or warmer (60F) on the other. What it ignores is how the drinking is done. Let's take some likely heat-stressed athletes which would be outdoors meaning ignoring indoor types operating with nice HVAC. You have a very different situation comparing cyclists sipping some COLD water as they want on the one hand and a marathoner or futbaler grabbing a cup or two of ice-cold water and chugging it down in a few seconds when it is offered/available. In the former situation, the cyclists are not going to significantly cool their stomachs. The internal cooling effect is going to be spread out between their mouth, esophagus and further down their guts. In the latter situation, you have that local gut cooling/slowing problem that could be real.
There could be good data out there that illuminates this problem, but I did not find it in a quick search. FWIW, most of what I just typed is just hand-waving as well, but at least some of it is quantitative hand-waving and thanks to Jim for starting us down the numbers path. My science motto is, "If you can't be quantitative F*** it".
Let's round things a little and say that body temp is 100F and you are considering drinking water at 40F or 70F. Water has much of the heat capacity in a body. The other stuff has some, but not nearly as much as water which is one of the unusual qualities of that substance. A person of 167# has 100# of water. If your 167# drinks a pint of 40F or 70F of water, you will lower your body temp by roughly 0.6 or 0.3 degrees respectively assuming total and immediate "mixing" That is much more significant that I might have assumed before looking at it quantitatively.
I am however, disturbed by the lack of detail that I find in discussions by people who project as experts in this respect. We are not just a bag of water and some other stuff being diluted with a little bag of water.
I have a vague recollection of some expert stating that drinking cold water slows absorption. It makes sense that cold water makes your gut cold and slows everything down at best and shuts it down at worst. I just went with that in my previous post, shame on lazy me. Local drastic cooling, however, just slows or stops everything that an organ is doing and that includes absorbing water. In a biology laboratory, we make processes of all kinds stop, or nearly stop by putting them in and ice-water bath. (That said, it is not as effective with poiklothermic cells/tissues/animals.)
I am not trying to refute that "cold stunned gut" reasoning, but I find little data to support it. There is data that supports drinking cold or cool water on the one hand, or warmer (60F) on the other. What it ignores is how the drinking is done. Let's take some likely heat-stressed athletes which would be outdoors meaning ignoring indoor types operating with nice HVAC. You have a very different situation comparing cyclists sipping some COLD water as they want on the one hand and a marathoner or futbaler grabbing a cup or two of ice-cold water and chugging it down in a few seconds when it is offered/available. In the former situation, the cyclists are not going to significantly cool their stomachs. The internal cooling effect is going to be spread out between their mouth, esophagus and further down their guts. In the latter situation, you have that local gut cooling/slowing problem that could be real.
There could be good data out there that illuminates this problem, but I did not find it in a quick search. FWIW, most of what I just typed is just hand-waving as well, but at least some of it is quantitative hand-waving and thanks to Jim for starting us down the numbers path. My science motto is, "If you can't be quantitative F*** it".
The engineer I was speaking of said it to me on a hot summer day while we were baking in the sun while standing on about 20 acres of a heavily packed clay soil being spread over a closed landfill. We were driving tubes into the clay soil to pluck out samples at random locations. He kept a gallon of water in a jug in the vehcle he drove and offered me some. I declined. Nothing but cold water for me Buster Brown.
We were both in our early 20's. By the time I reached my 30's I was using his method to a certain degree. I'd keep a pair of quart Gatorade bottles filled with water in the cab of my truck and a pair frozen as ice blocks in my lunch pale to keep my lunch cool. After lunch I'd let them melt and drink the water from those. Back then I also drank a lot of beer after work hours.
In my 40's I no longer drank any beer at all. My body started telling me to drink less coffee, more water and by all means add some of the stuff that comes out when sweating. Did I listen? Of course not. Not until a few trips to the ER anyway, but even then it really didn't sink in until my 50's. That's when the muscle cramps began. Not leg cramps. Torso cramps like shoulders and abdomen.
In warm weather I store gallons of water in my truck and drink it at "cabin" temperature meaning whatever temperature the cab of my truck happens to be. I sill keep frozen water as ice blocks for my lunch pale but rarely consume that water. Partly because the water has been stored in them for a few years now and partly because it's heavily salted to freeze harder but if worse comes to worse... it is available. In winter I carry a daily supply since often the stockpile stored in the truck ends up frozen.
The water helps thwart the cramps when they happen. A diet rich in electrolytes helps prevent them in the first place.
We were both in our early 20's. By the time I reached my 30's I was using his method to a certain degree. I'd keep a pair of quart Gatorade bottles filled with water in the cab of my truck and a pair frozen as ice blocks in my lunch pale to keep my lunch cool. After lunch I'd let them melt and drink the water from those. Back then I also drank a lot of beer after work hours.
In my 40's I no longer drank any beer at all. My body started telling me to drink less coffee, more water and by all means add some of the stuff that comes out when sweating. Did I listen? Of course not. Not until a few trips to the ER anyway, but even then it really didn't sink in until my 50's. That's when the muscle cramps began. Not leg cramps. Torso cramps like shoulders and abdomen.
In warm weather I store gallons of water in my truck and drink it at "cabin" temperature meaning whatever temperature the cab of my truck happens to be. I sill keep frozen water as ice blocks for my lunch pale but rarely consume that water. Partly because the water has been stored in them for a few years now and partly because it's heavily salted to freeze harder but if worse comes to worse... it is available. In winter I carry a daily supply since often the stockpile stored in the truck ends up frozen.
The water helps thwart the cramps when they happen. A diet rich in electrolytes helps prevent them in the first place.
PhotonWrangler
Flashaholic
So I've been having leg cramps at night for awhile. For the past two nights I tried eating a couple of pickle slices an hour or so before bed, and no leg cramps. It's too soon for me to know if this is real or a coincidence but I will continue with pickles or pickle brine for awhile and see how it goes.
- Joined
- Mar 4, 2012
- Messages
- 643
Which is why pure water doesn't cut it. Without electrolytes you can put as much H2O in you as you can afford and it still won't help or satisfy.
Jim must've meant a very diluted saltwater mix in the vein of a saline solution.
Yes, I meant just enough salt (e.g. the only electrolyte that you can replace in minutes) to replace what you lose to sweat. I actually will come in from my warehouse and put a little salt in my coffee cup (1000mg), add a little hot water, and drink it down. I'm also drinking 5 big swallows or so of room temp water very 15 minutes. That's about the most you can process over time.
I can only say this works for me. I rode 40 miles of sandy whoops on Friday. It was a "feels like" 104 degree day. I was in motocross boots, socks, knee brace sleeves, full knee braces, MX pants, hard armor long sleeve jacket with full turtleback, Ogio flight vest, gloves, helmet, and goggles. Then I ran out of gas with about 300 yards of deep sand trail left. First time in 52 years of riding dirt bikes! Pushed the bike back to the parking lot. No problem.
Working in my warehouse is cheating, because my body is majorly heat adapted (a real thing...Google it). It's generally 10 degrees hotter in the warehouse than outside, but almost as humid.
Last edited:
PhotonWrangler
Flashaholic
This has worked for me in the past. I need to get another bottle of them.
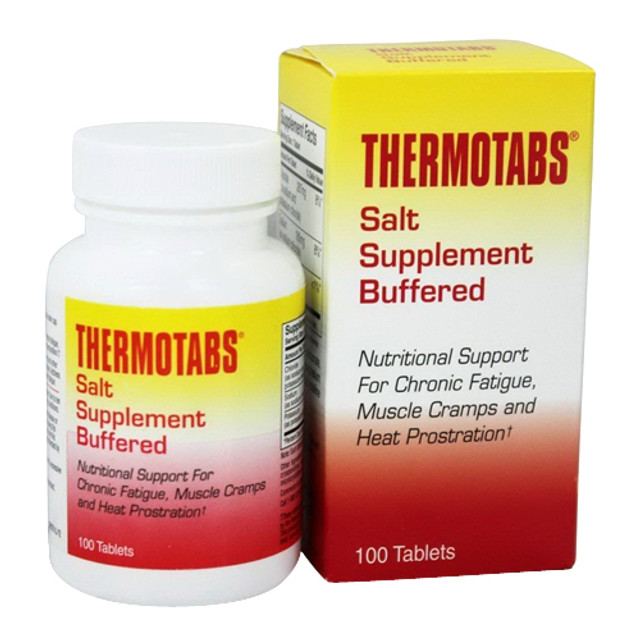
- Joined
- Mar 4, 2012
- Messages
- 643
That's what my 98 year old mother in law takes. I use unbuffered just because it's cheap and I have no problem tolerating the taste long enough to swallow one pill.
knucklegary
Flashlight Enthusiast
Salt creates water retention. Not recommended for people who are overweight with heart issues?That's what my 98 year old mother in law takes. I use unbuffered just because it's cheap and I have no problem tolerating the taste long enough to swallow one pill.
- Joined
- Mar 4, 2012
- Messages
- 643
Salt creates water retention. Not recommended for people who are overweight with heart issues?
Excess salt can be unhealthy. My mil was consistently low, so she supplements per her doctor.
You will die without salt. Cannot live without it. I'm replacing lost electrolyte to restore a healthy balance. See how cool it sounds when you call it electrolyte? Salt (the sodium portion) is the only electrolyte that you lose in sweat that can be replaced in minutes. The others take hours to days, so I replace those with diet or supplementation over time.
alpg88
Flashlight Enthusiast
- Joined
- Apr 19, 2005
- Messages
- 5,368
Too much salt does not let kidneys remove water from blood, and regulate blood pressure, not much of an issue with healthy people, whose arteries are still soft and flexible, but for those whose are hardened, and do have high blood pressure, it can cause problems. OTOH dehydration makes blood thicker, another problem for people with hardened arteries especially if they are narrowed due to plaque. aspirin helps to keep blood thinner even if dehydrated. worst thing is, lots of people do not know they have narrowed arteries, no one gets a stress test, or an Electronic angiogram unless they have symptoms, symptoms start when arteries are narrowed more than 60% some do not feel it until they are almost blocked. My coworker always felt great, then 1 day during his morning run, he passed out, woke up at the hospital, one of his arteries was 90% blocked.Salt creates water retention. Not recommended for people who are overweight with heart issues?
